Aluminum is a silvery-white lightweight metal of main group 13 from the periodic table. It is the most widely used non-ferrous metal. Let us discuss the engineering properties, production, and different alloys of aluminum.

Engineering Properties of Aluminum
Aluminum possesses a combination of properties that make it an extremely useful engineering material.
- Aluminum has a low density (2.70 g/cm3 ), making it particularly useful for transportation manufactured products.
- Aluminum also has good corrosion resistance in most natural environments due to the tenacious oxide film that forms on its surface.
- Although pure aluminum has low strength, it can be alloyed to a strength of about 100 ksi (690 MPa).
- Aluminum is nontoxic and is used extensively for food containers and packaging.
- The good electrical properties of aluminum make it suitable for many applications in the electrical industry.
- The relatively low price of aluminum along with its many useful properties make this metal very important for industrially.
Elemental properties of Aluminum
| Element Properties | |
| Atomic number | 13 |
| Atomic weight | 26.9815384 |
| Melting point | 660 °C (1,220 °F) |
| Boiling point | 2,467 °C (4,473 °F) |
| Specific gravity | 2.70 (at 20 °C [68 °F]) |
| Valence | 3 |
| Electron configuration | 1s22s22p63s23p1 |
Production of Aluminum
Aluminum is the most abundant metallic element in the earth’s crust and always occurs in the combined state with other elements such as iron, oxygen, and silicon. Bauxite, which consists mainly of hydrated aluminum oxides, is the chief commercial mineral used for the production of aluminum.
- In the Bayer process, bauxite is reacted with hot sodium hydroxide to convert the aluminum in the ore to sodium aluminate.
- After the separation of the insoluble material, aluminum hydroxide is precipitated from the aluminate solution.
- The aluminum hydroxide is then thickened and calcined to aluminum oxide, Al2O3.
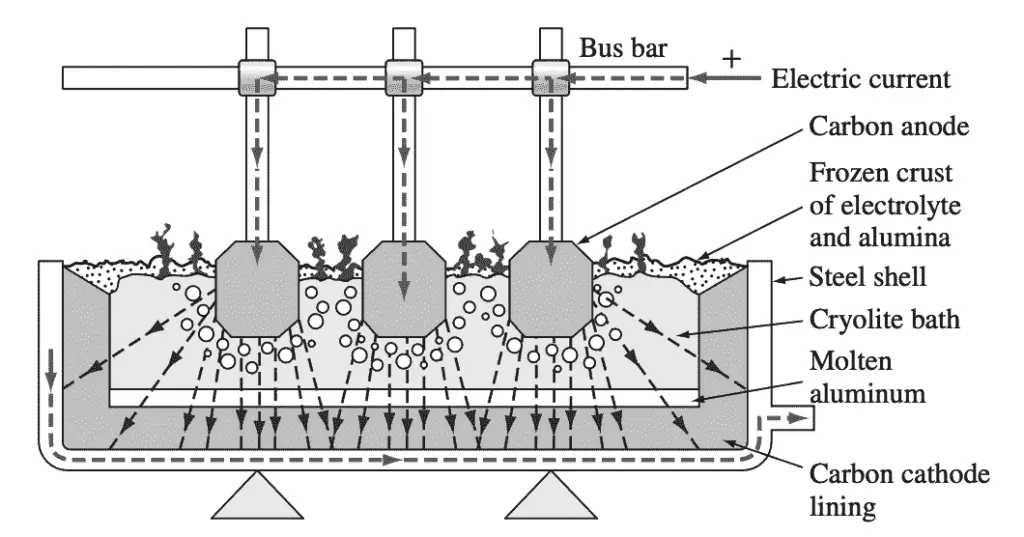
- The aluminum oxide is dissolved in a molten bath of cryolite (Na3AlF6) and electrolyzed in an electrolytic cell by using carbon anodes and cathodes.
- In the electrolysis process, metallic aluminum forms in the liquid state and sinks to the bottom of the cell and is periodically tapped off.
- The cell-tapped aluminum usually contains from 99.5% to 99.9% aluminum with iron and silicon being the major impurities.
- Aluminum from the electrolytic cells is taken to large refractory-lined furnaces, where it is refined before casting.
- Alloying elements and alloying-element master ingots may also be melted and mixed in with the furnace charge.
- In the refining operation, the liquid metal is usually purged with chlorine gas to remove dissolved hydrogen gas, which is followed by skimming off the liquid-metal surface to remove oxidized metal.
- After the metal has been degassed and skimmed, it is screened and cast into ingot shapes for remelting or into primary ingot shapes such as sheet or extrusion ingots for further fabrication.
Primary Fabrication of Aluminum
Ingot shapes such as sheet and extrusion ingots are usually semi-continuously cast by the direct-chill method.
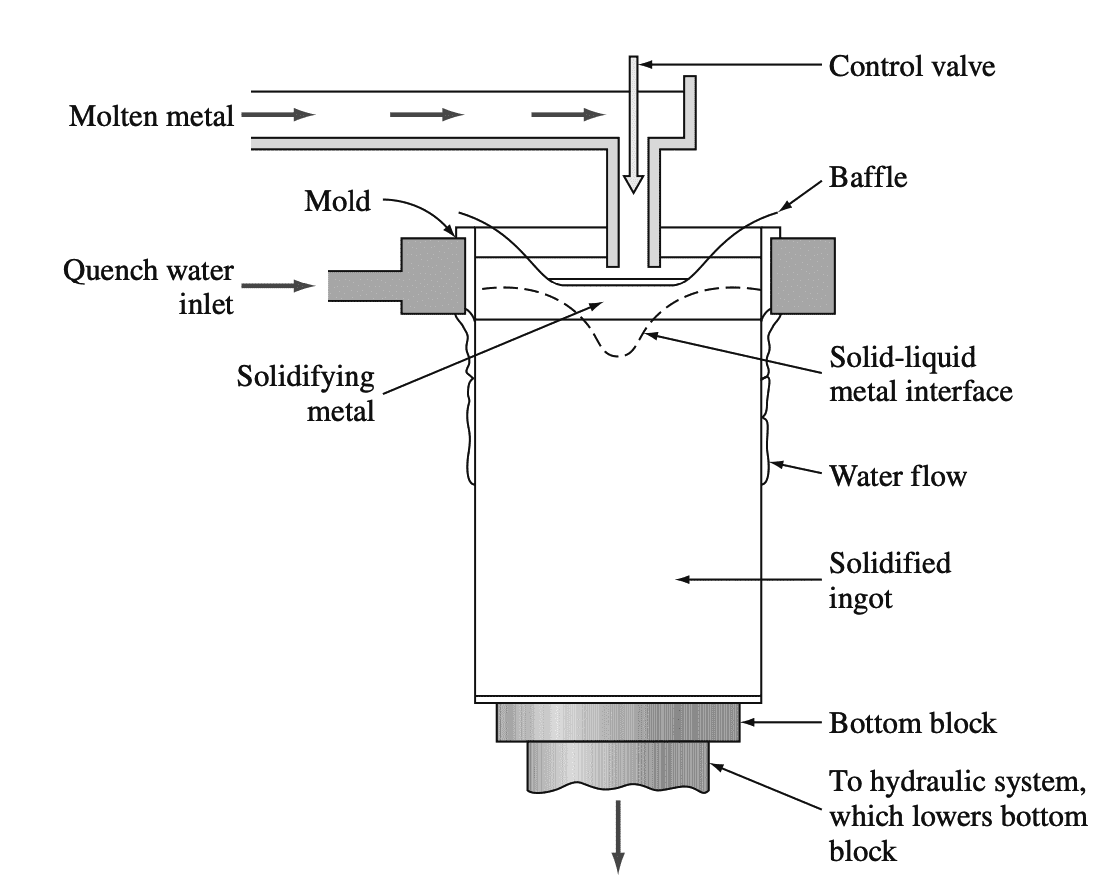
Above schematic shows schematically how an aluminum ingot is cast by this method.
- In the case of sheet ingots, about 1/2 in. of metal is removed from the ingot surfaces that will make contact with the hot-rolling-mill rolls.
- This operation is called scalping and is done to ensure a clean, smooth surface for the fabricated sheet or plate.
- Next, the ingots are preheated or homogenized at a high temperature for about 10 to 24 h to allow atomic diffusion to make the composition of the ingot uniform.
- The preheating must be done at a temperature below the melting point of the constituent with the lowest melting temperature.
- After reheating, the ingots are hot-rolled by using a four-high reversing hot-rolling mill.
- The ingots are usually hot-rolled to about 3 in. thick and then reheated and hot-rolled down to about 3/4 to 1 in. With an intermediate hot-rolling mill.
- Further reduction is usually carried out on a series of tandem hot-rolling mills to produce metal about 0.1 in. thick.
- Following figure shows a typical cold-rolling operation. More than one intermediate anneal is usually required if the thin sheet is to be produced.

Classification of Wrought Aluminum Alloys
Aluminum alloys produced in the wrought form i.e., sheet, plate, extrusions, rod, and wire are classified according to the major alloying elements they contain.
A four-digit numerical designation is used to identify aluminum wrought alloys.
- The first digit indicates the alloy group that contains specific alloying elements.
- The last two digits identify the aluminum alloy or indicate the aluminum purity.
- The second digit indicates modification of the original alloy or impurity limits.
The followiung table 1 lists the wrought aluminum alloy groups.
Table 1: Wrought aluminum alloy groups
| Alloy | Group |
| Aluminum, 99.00% minimum and greater | 1xxx |
| Aluminum alloys grouped by major alloying elements: | |
| Copper | 2xxx |
| Manganese | 3xxx |
| Silicon | 4xxx |
| Magnesium | 5xxx |
| Magnesium and silicon | 6xxx |
| Zinc | 7xxx |
| Other element | 8xxx |
| Unused series | 9xxx |
Temper Designations
Temper designations for wrought aluminum alloys follow the alloy designation and are separated by a hyphen (for example, 1100-0). Subdivisions of a basic temper are indicated by one or more digits and follow the letter of the basic designation (for example, 1100-H14).
Basic Temper Designations
- F — As fabricated. No control over the amount of strain hardening; no mechanical property limits.
- O — Annealed and recrystallized. Temper with the lowest strength and highest ductility.
- H — Strain-hardened (see subsequent subsection for subdivisions).
- T — Heat-treated to produce stable tempers other than F or O (see subsequent subsection for subdivisions).
Strain-Hardened Subdivisions
- H1—Strain-hardened only. The degree of strain hardening is indicated by the second digit and varies from quarter-hard (H12) to full-hard (H18), which is produced with an approximately 75% reduction in area.
- H2—Strain-hardened and partially annealed. Tempers ranging from quarter hard to full-hard obtained by partial annealing of cold-worked materials with strengths initially greater than desired. Tempers are H22, H24, H26, and H28.
- H3—Strain-hardened and stabilized. Tempers for age-softening aluminum–magnesium alloys that are strain-hardened and then heated at a low temperature to increase ductility and stabilize mechanical properties. Tempers are H32, H34, H36, and H38.
Heat-Treated Subdivisions
- T1—Naturally aged. The product is cooled from an elevated temperature shaping process and naturally aged to a substantially stable condition.
- T3—Solution heat-treated, cold-worked, and naturally aged to a substantially stable condition.
- T4—Solution heat-treated and naturally aged to a substantially stable condition.
- T5—Cooled from an elevated temperature shaping process and then artificially aged.
- T6—Solution heat-treated and then artificially aged.
- T7—Solution heat-treated and stabilized.
- T8—Solution heat-treated, cold-worked, and then artificially aged
Non–Heat-Treatable Wrought Aluminum Alloys
Wrought aluminium alloys can conveniently be divided into two groups: non–heat-treatable and heat-treatable alloys.
Non–heat-treatable aluminium alloys cannot be precipitation-strengthened but can only be cold-worked to increase their strength. The three main groups of non–heat-treatable wrought aluminium alloys are the 1xxx, 3xxx, and 5xxx groups.
Following Table 2 lists the chemical composition, typical mechanical properties, and applications for some selected industrially important wrought aluminium alloys.
Table 2: Typical mechanical properties and applications for Wrought aluminum alloys
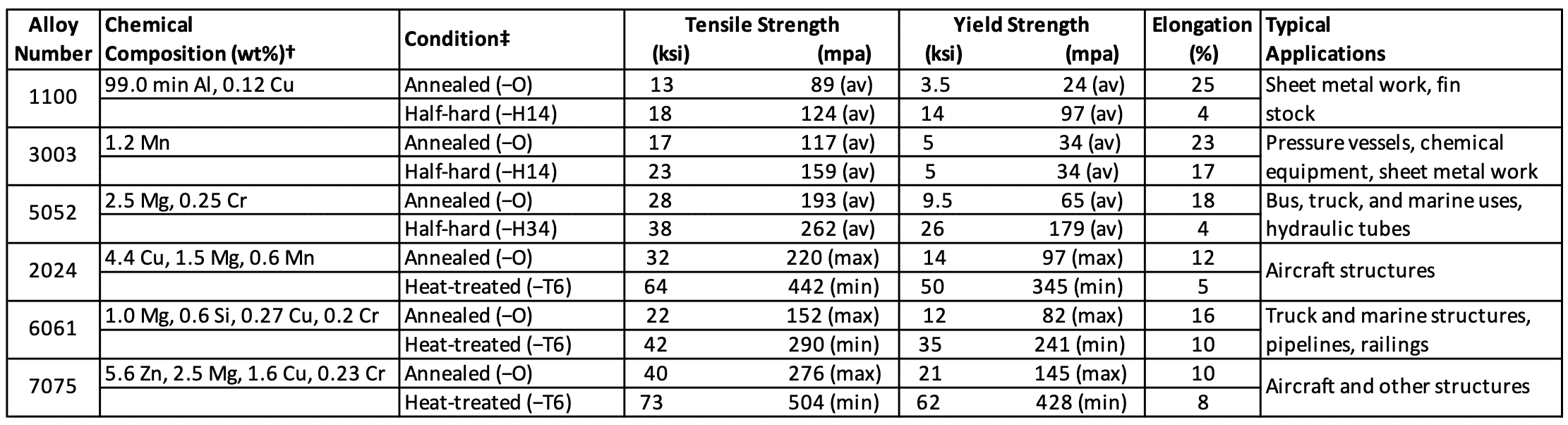
- 1xxx alloys: These alloys have a minimum of 99.0% aluminium, with iron and silicon being the major impurities (alloying elements). An addition of 0.12% copper is added for extra strength. The 1100 alloy has a tensile strength of about 13 ksi (90 MPa) in the annealed condition and is used mainly for sheet metal work applications.
- 3xxx alloys: Manganese is the principal alloying element of this group and strengthens aluminium mainly by solid-solution strengthening. The most important alloy of this group is 3003, which is essentially an 1100 alloy with the addition of about 1.25% manganese. The 3003 alloy has a tensile strength of about 16 ksi (110 MPa) in the annealed condition and is used as a general-purpose
alloy where good workability is required. - 5xxx alloys: Magnesium is the principal alloying element of this group and is added for solid-solution strengthening in amounts up to about 5%. One of the most industrially important alloys of this group is 5052, which contains about 2.5% magnesium (Mg) and 0.2% chromium (Cr). In the annealed condition, alloy 5052 has a tensile strength of about 28 psi (193 MPa). This alloy is also used for sheet metal work, particularly for bus, truck, and marine applications.
Heat-Treatable Wrought Aluminum Alloys
Some aluminium alloys can be precipitation-strengthened by heat treatment. Heat-treatable wrought aluminium alloys of the 2xxx, 6xxx, and 7xxx groups are all precipitation-strengthened by similar mechanisms as described previous article for aluminium-copper alloys.
The above Table 2 lists the chemical compositions, typical mechanical properties, and applications of some of the industrially important wrought heat-treatable aluminium alloys.
- 2xxx alloys: The principal alloying element of this group is copper, but magnesium is also added to most of these alloys. Small amounts of other elements are also added. One of the most important alloys of this group is 2024, which contains about 4.5% copper (Cu), 1.5% Mg, and 0.6% Mn. This alloy is strengthened mainly by solid-solution and precipitation strengthening. An intermetallic compound of the approximate composition of Al2CuMg is the main strengthening precipitate. Alloy 2024 in the T6 condition has a tensile strength of about 64 ksi (442 MPa) and is used, for example, for aircraft structural.
- 6xxx alloys: The principal alloying elements for the 6xxx group are magnesium and silicon, which combine to form an intermetallic compound, Mg2Si, which in precipitate form strengthens this group of alloys. Alloy 6061 is one of the most important alloys of this group and has an approximate composition of 1.0% Mg, 0.6% Si, 0.3% Cu, and 0.2% Cr. This alloy in the T6 heat-treated condition has a tensile strength of about 42 psi (290 MPa) and is used for general-purpose structurals.
- 7xxx alloys: The principal alloying elements for the 7xxx group of aluminium alloys are zinc, magnesium, and copper. Zinc and magnesium combine to form an intermetallic compound, MgZn2, which is the basic precipitate that strengthens these alloys when they are heat-treated. The relatively high solubility of zinc and magnesium in aluminium makes it possible to create a high density of precipitates and hence to produce very great increases in strength. Alloy 7075 is one of the most important alloys of this group; it has an approximate composition of 5.6% Zn, 2.5% Mg, 1.6% Cu, and 0.25% Cr. Alloy 7075, when heat-treated to the T6 temper has a tensile strength of about 73 psi (504 MPa) and is used mainly for aircraft structurals.
Aluminum Casting Processes
Aluminium alloys are normally cast by one of three main processes:
- Sand casting
- Permanent mould
- Die casting
1. Sand casting
Sand casting is the simplest and most versatile of the aluminium casting processes. Following figure shows how a simple sand mould for producing sand castings is constructed.
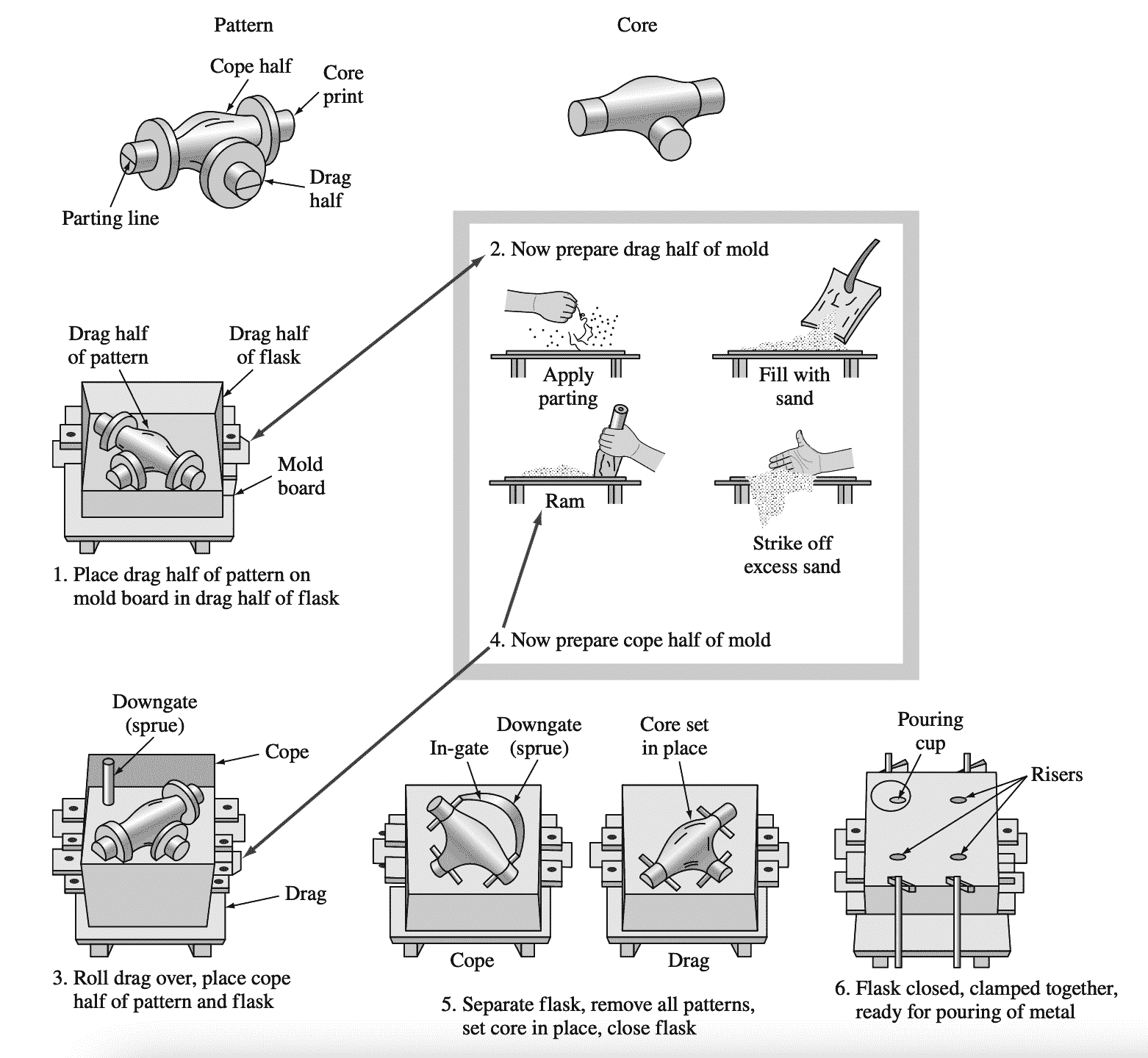
Steps in the construction of a simple sand mould for making a sand casting
The sand-casting process is usually chosen for the production of
(1) small quantities of identical castings,
(2) complex castings with intricate cores,
(3) large castings
(4) structural castings
Permanent mould
In permanent-mould casting, the molten metal is poured into a permanent metal mould under gravity, low pressure, or centrifugal pressure only.

The above figure shows an open permanent mould and the permanent-mould casting of aluminium alloy components.
Castings of the same alloy and shape produced by a permanent mould have a finer grain structure and higher strength than those cast by sand moulds. The faster cooling rate of permanent-mould casting produces a finer grain structure. Also, permanent-mould castings usually have less shrinkage and gas porosity than sand castings. However, permanent moulds have size limitations, and complex parts may be difficult or impossible to cast with a permanent mould.
Die Casting
In die casting, identical parts are cast at maximum production rates by forcing molten metal under considerable pressure into metal moulds.

Two metal die halves are securely locked together to withstand high pressure. The molten aluminium is forced into the cavities in the dies. When the metal has solidified, the dies are unlocked and opened to eject the hot casting. The die halves are locked together again, and the casting cycle is repeated.
Some of the advantages of die casting are
- Parts die cast are almost completely finished and can be produced at high rates,
- Dimensional tolerances of each cast part can be more closely held than with any other major casting process,
- Smooth surfaces on the casting are obtainable,
- Rapid cooling of the casting produces a fine-grain structure,
- The process can be automated easily
Read more about casting processes here.
Aluminum Casting Alloy Compositions
Aluminium casting alloys have been developed for casting qualities such as fluidity and feeding ability as well as for properties such as strength, ductility, and corrosion resistance. As a result, their chemical compositions differ greatly from those of the wrought aluminium alloys.
Table 3: Typical mechanical properties and applications for Cast aluminum alloys
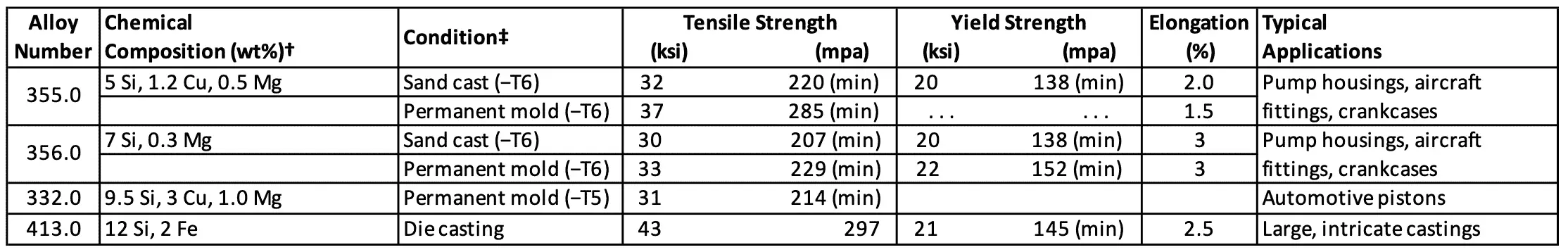
The above Table 3 lists the chemical compositions, mechanical properties, and applications for some selected aluminium casting alloys. These alloys are classified in the United States according to the Aluminum Association system. In this system, aluminium casting alloys are grouped by the major alloying elements they contain by using a four-digit number with a period between the last two digits, as listed in Table 4.
Table 4: Cast aluminum alloy groups
| Alloy | Group |
| Aluminum, 99.00% minimum and greater | 1xx.x |
| Aluminum alloys grouped by major alloying elements: | |
| Copper | 2xx.x |
| Silicon, with added copper and/or magnesium | 3xx.x |
| Silicon | 4xx.x |
| Magnesium | 5xx.x |
| Zinc | 7xx.x |
| Tin | 8xx.x |
| Other element | 9xx.x |
| Unused series | 6xx.x |
- Silicon in the range of about 5% to 12% is the most important alloying element in aluminium casting alloys since it increases the fluidity of the molten metal and its feeding ability in the mould and also strengthens the aluminium.
- Magnesium in the range of about 0.3% to 1% is added to increase strength, mainly by precipitation strengthening through heat treatment.
- Copper in the range of about 1% to 4% is also added to some aluminium casting alloys to increase strength, particularly at elevated temperatures.
- Other alloying elements such as zinc, tin, titanium, and chromium are also added to some aluminium casting alloys.
In some cases, if the cooling rate of the solidified casting in the mould is sufficiently rapid, a heat-treatable alloy can be produced in the supersaturated solid condition. Thus, the solution heat-treatment and quenching steps can be omitted for precipitation strengthening the casting, and only subsequent ageing of the casting after it has been removed from the mould is required.
A good example of the application of this type of heat treatment is in the production of precipitation-strengthened automobile pistons.

The components are shown in above figure, after being removed from the mould, only require an ageing treatment to be precipitation-strengthened. This heat-treatment temper is called T5.
Conclusion
We have discussed Engineering Properties of Aluminum, Production & Fabrication of Aluminum, Classification of Wrought Aluminum Alloys, Aluminum Casting Alloy Compositions in detail, let us know what else we are missing about aluminum in the comment section below.

You have made valid points in a unique way. As a result of reading this article, I will be better informed Production of Aluminum and Aluminum Alloys
Great. Thanks