We have discussed the different air standard cycles in the previous articles, Lenoir Cycle is one of the idealized thermodynamic Air standard cycles which is used in the Pulse Jet Engines.
Lenoir Cycle
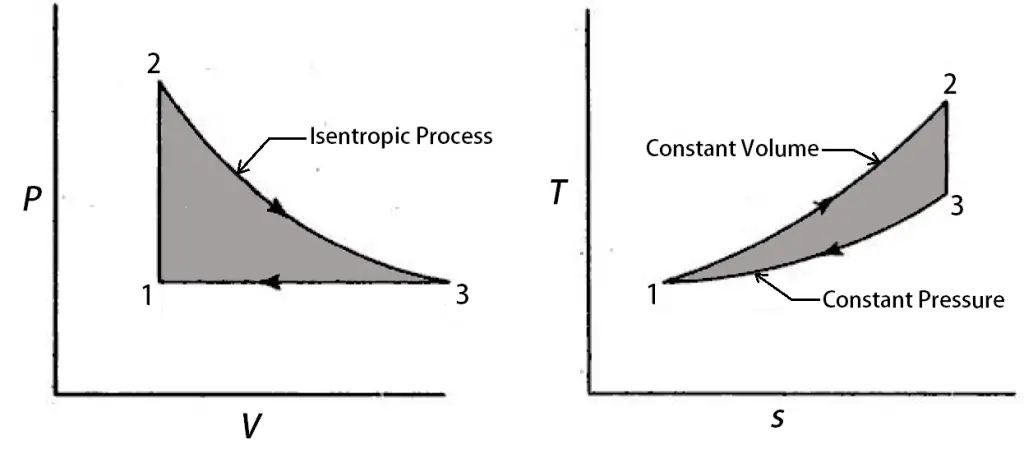
Lenoir Cycle consists of a constant volume heat addition, Isentropic expansion, and the constant pressure heat rejection processes. There is no compression process for the working fluid.
Due to the absence of the compression process, it will results in less thermal efficiency than the other Air-Standard Cycles such as the Otto Cycle, Diesel Cycle, Dual Cycle.
Check the P-V diagram and the T-S diagram for the Lenoir Cycle below.
1 → 2
1 → 2 is a Constant Volume heat addition process. Where the fuel will be burnt under a constant volume.
2 → 3
2 → 3 is an Isentropic Expansion, right after the combustion happens. It is a
reversible adiabatic expansion process. Actually where the work is generated.
3 → 1
3 → 1 is the Constant Pressure heat rejection, where the combustion particles will be released into the surroundings at a constant pressure.
Thermal Efficiency of Lenoir Cycle
Thermodynamically, the efficiency is given by
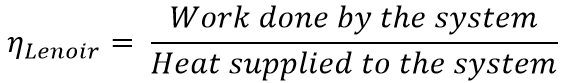
Work done by the system = Heat supplied (QS)- Heat rejected (QR)
W = QS – QR
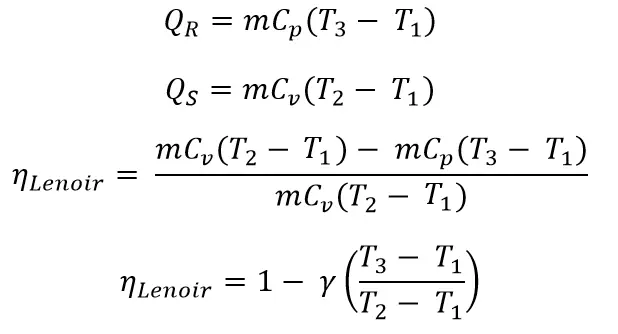
From process 1 → 2
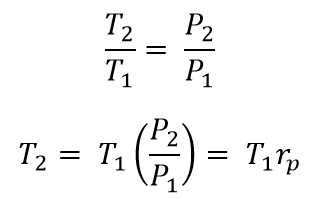
Where
From Process 2 → 3
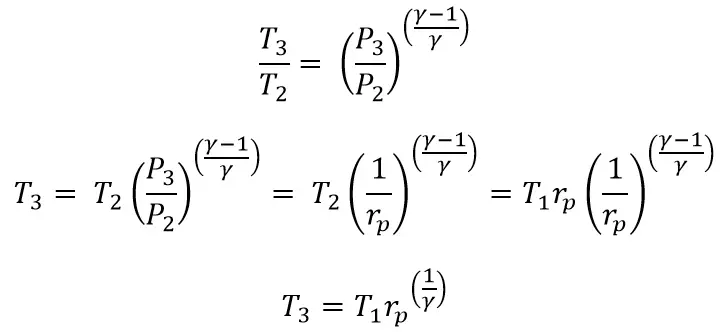
Let’s substitute these two above equations in the main efficiency equation.
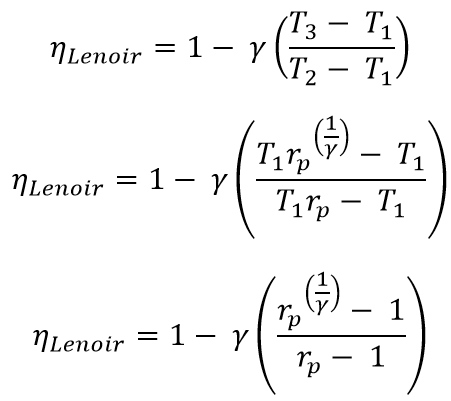
From the above efficiency equation, we can observe that the efficiency of the Lenoir Cycle is dependent on the pressure ratio, so It is poor in the thermal efficiency when we compared to the thermal efficiencies of the
Otto Cycle, Diesel Cycle, Dual Cycle.
This Lenoir cycle is used in the Pulse Jet engines only.
Conclusion
We have discussed the different processes in the Lenoir Cycle with the P-V and T-S diagrams and also we have derived the equation for the Efficiency of this cycle. If you have any thoughts on this topic, Please let us know in the comment section below.

Leave a Reply